
The C-O-C linkage can also undergo a symmetric stretch as shown in the right side of Figure 2. This vibration is analogous to the C-C-O asymmetric stretch of alcohols, and like alcohols these peaks are typically the largest ones found between 13 cm -1 (for the rest of this installment assume all peak positions quoted are in cm -1 units if not specified). Since all ethers contain a C-O-C linkage, the vibration that we would usually refer to as a “C-O” stretch actually involves the asymmetric stretch of the C-O-C moiety as shown in the left side of Figure 2. If both ether carbons are saturated then we have a saturated ether, if both ether carbons are aromatic that gives an aromatic ether, and if one ether carbon is saturated and one is aromatic it is a mixed ether. 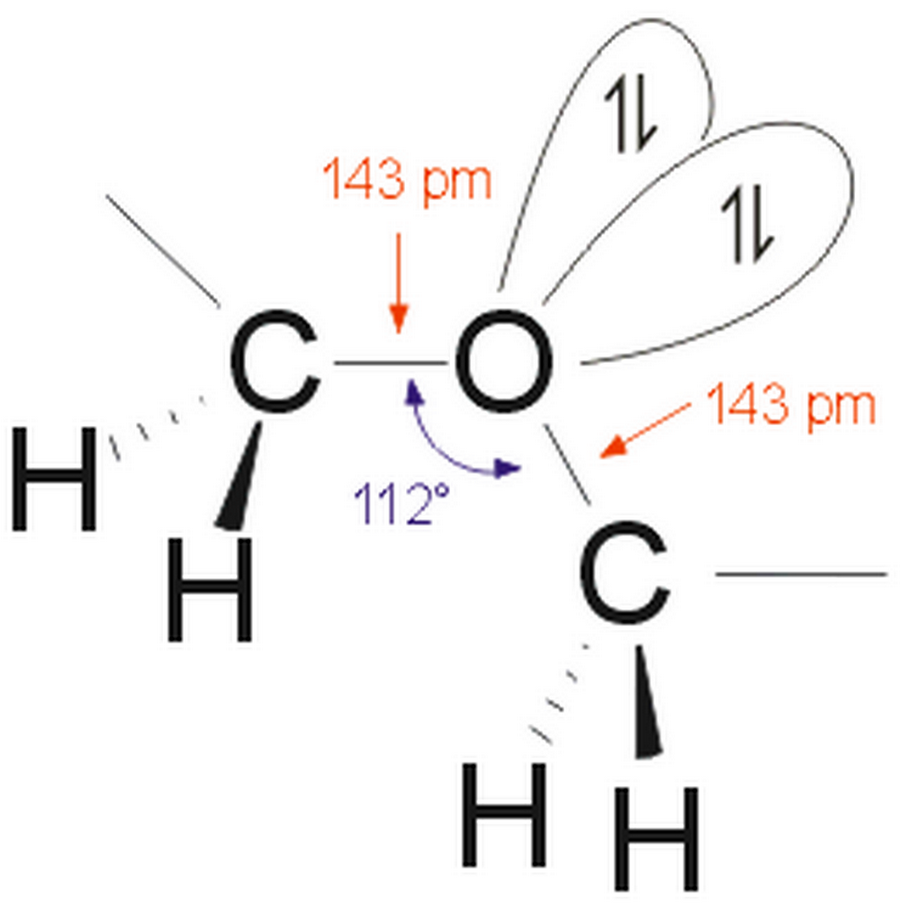 
Here we discuss three different types of ether, which are characterized by the type of carbons attached to the central oxygen. In fact, the only useful group wavenumbers for ethers are their C-O stretching peaks as discussed below. Because ethers do not contain an -OH bond there is no significant hydrogen bonding and there are no -OH stretching or bending peaks. The carbons in these bonds are referred to as ether carbons as shown in the figure. Note that unlike alcohols ethers contain not one but two C-O bonds. Ethers are characterized by a central oxygen atom with two carbons attached, as shown in Figure 1. Now that we are finished with these molecules the next functional group to study that contains the C-O bond is ethers. We discuss three different types of ether, which are characterized by the type of carbons attached to the central oxygen.Ī few issues ago we started our survey of the infrared (IR) spectroscopy of the C-O bond (1), and proceeded to study the spectra of alcohols and phenols (1,2).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
